Background:
In the ongoing phase 3 JAVELIN Renal 101 trial, progression-free survival (PFS) was longer (median, 13.8 vs 8.4 mo; hazard ratio, 0.69; p=0.0001) and the objective response rate (ORR) was higher (51% vs 26%) with A + Ax vs S in patients with previously untreated aRCC. Here we report outcomes from an analysis of several prespecified subgroups.
Methods:
Patients were randomized 1:1 to receive A (10 mg/kg) IV every 2 weeks + Ax (5 mg) PO twice daily or S (50 mg) PO once daily for 4 wk (6-wk cycle). Primary and key secondary endpoints were PFS per independent review committee (IRC; RECIST v1.1) and OS in patients with PD-L1+ tumors (≥1% of immune cells) and in patients irrespective of PD-L1 expression; other secondary endpoints included OR per IRC (RECIST v1.1).
Results:
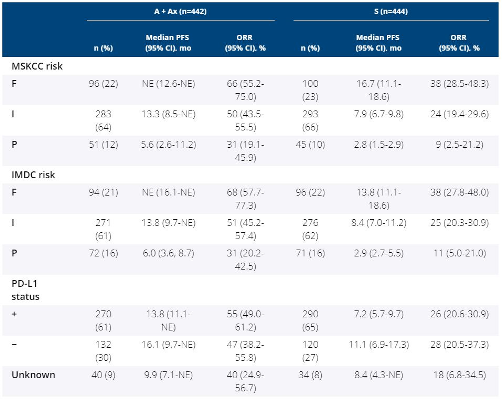
A total of 886 patients were randomized; 560 (63%) had PD-L1+ tumors. At data cut-off (Jun 2018), median follow-up was 12.0 vs 11.5 mo for A + Ax vs S groups. The table shows PFS and ORR by MSKCC and IMDC risk groups (F, favorable; I, intermediate; P, poor) and PD-L1 subgroup. Similar results for prognostic risk were seen in patients with PD-L1+ tumors. Outcome data (including PFS2) for additional clinical subgroups by baseline demographics and features will be presented. Clinical trial information: NCT02684006
Conclusions:
A + Ax demonstrated PFS and OR benefit across all prognostic risk groups and PD-L1 subgroups vs S in aRCC.
NE
, not estimable